
The history of medical imaging has included a number of
different imaging systems and innovators, but it is fairly
clear that the story really starts with Wilhelm Rontgen
(1845-1923).
In 1895, Wilhelm Rontgen was experimenting with discharging
electrical current in 'Crookes' tubes, when he noticed that a
barium platinocyanide coated screen across the room was
glowing, despite the tube being encased in cardboard.
Whilst experimenting with holding items between the tube and
the screen, he soon found that the mysterious rays would pass
through his flesh, but cast an outline of his bones on the
screen. These findings were published in the Wurzburg
Physical-Medical Journal, and as the world realised the
medical value of the X-ray, Rontgen was awarded the first
Nobel prize for physics in 1901.
The X-ray went on to become a vital tool to the medical
profession (although its dangers were not appreciated until
much later), but the next steps in internal medical imaging
were not made until the end of World War II.
The development of magnetic resonance is attributed to the
work of Felix Bloch and Edward Purcell in 1946, who
independently discovered the presence of magnetic resonance
in solids and liquids. They were able to create devices that
would show images of these fields, and they were awarded the
Nobel prize in 1952 for their work.
During the period 1950-1970, nuclear magnetic resonance was
developed for chemical and physical molecular analysis,
rather than medical applications. In 1967, the next
developments in NMR were made by Paul Lauterbur, who found
that a photographic-style image could be generated using
magnetic resonance (previously this had only been possible
using light and x-rays).
At about the same time, a physician named Raymonde Damadian
discovered that malignant body tissue had a different
spectrum than normal tissue. In 1974 he was able to produce a
crude image of a tumor in a rat, and in 1976 he produced an
image of the human body which took four hours to complete.
These early MRI scanners were able to display results in
tomographic layers, and would certainly have been impossible
to construct without the development of digital computers and
electronics.
Meanwhile, a different medical imaging system had been
developed by British engineer Godfrey Hounsfield of EMI
Laboratories. Hounsfield invented Computed Tomography(CT)
imaging in 1972. This scanning technique involves combining
x-ray images from a detector that rotates 360 degrees around
the subject. A dedicated computer extrapolates this
information into a two-dimensional image of the scanned
'slice'.
The first CT scanners were installed between 1974 and 1976,
becoming widely available by 1980. The main importance of
this was that it showed that hospitals were willing to pay
for expensive medical imaging machines. The first CT scanner
took several hours to aquire the raw data for a 'slice', and
days to reconstruct a single image from this data! Increases
in technology have decreased the time taken for a scan, with
a modern CT scanner able to scan a 'slice' in about 100ms,
and reconstruct an image in less than a second.
In parallel to the establishment of CT scanners in general
use, Damadian continued his work, setting up a company to
sell his MRI medical imaging machines. The next major
innovation in the field of MRI was made by Dumoulin, who
perfected MRI angiography in 1988. This scanning technique
allows imaging of flowing blood without the use of
contrasting agents. In 1989 echo-planar imaging was
introduced, which allowed image acquisition at video rates;
vital for the real-time systems which are being developed for
the future.
Very recent developments in the field of MRI (1999) have been
made in the development of image-guided neurosurgery, with an
open-magnet MRI machine designed by General Electric Medical
Systems and innovative software written by the AI laboratory
at Massachusetts Institute of Technology. The function of
this system is to take MRI scans and MRI angiograms, and
combine these to create a three dimensional model of the
patient's head and brain tissues, which is overlayed in
virtual reality over the surgeon's point of view as surgery
takes place.
X-rays are electromagnetic radiation ranging in wavelength from about 10 nm to 0.001 nm. They effect a photographic emulsion in the same way that light does. Absorption of X radiation by a substance depends upon its density and atomic weight. When the human body is x-rayed, the bones, which are composed of elements of higher atomic weight, absorb more radiation and cast darker shadows on the photographic plate.
Computed Tomography is based on the x-ray principle; as x-rays pass through the body they are absorbed or weakened at differing levels. Inside the CT machine is a semi-circular detector that measures the x-rays strength. This detector and the x-ray tube are mounted on a rotating frame that spins around the body of the patient taking roughly 1000 snapshots from different angles; every 360 degree rotation, a 'slice' is scanned. The x-ray tube may be focussed to a certain slice width (resolution) using lead shutters.
The software which drives the CT scanner has the complex task
of taking the 1000 x-ray images and backwards reconstructing
a two dimensional image of the 'slice' which was scanned.
There is a dedicated computer inside the scanner devoted to
this task of dealing with the raw CT data.
Magnetic resonance imaging is based on the absorption and emission of energy in the radio frequency range of the electromagnetic spectrum. The human body is primarily fat and water, and therefore contains approximately 63% hydrogen atoms. Magnetic resonance imaging primarily images the NMR signal from the hydrogen nuclei, which each comprise a single proton. The proton possesses a property called spin, which will cause the nucleus to produce an NMR signal.
In an MRI scanner, an enormous superconducting electromagnet (cooled by liquid helium) generates a powerful magnetic field. This field generates a steady state of hydrogen nuclei within the body. Within the magnet are gradient coils for producing a gradient in the X, Y and Z directions. Inside the gradient coils is the RF coil, which produces the magnetic pulse necessary to stimulate the hydrogen nuclei, and rotate the spins by 90 or 180 degrees. The RF coil also detects the signals from the spins within the body. (appendix 1).
The scan room must be shielded from external RF signals, to stop them from being picked up by the imager. A piece of equipment called a quadrature detector converts the signals returned from the RF coil into X and Y components. This data is fed to the computer, which builds up a cross-section image similar to that obtained from a CT scanner.
Once interpreted by the built-in algorithms, both CT and MRI machines produce two dimensional bitmap images of cross sections of the patient. One type of additional processing which software commonly does is the automatic separation and colour coding of tissue types. This is really a segmentation task that falls within the topic of computer vision.
The type of segmentation described in Image Guided Surgery is fairly simple; "each voxel must be labelled by tissue type and combined with like voxels into indentifiable structures." There isn't any explanation of how this would happen, but having read some books on segmentation of images (notably Boyle/Thomas), I think that it is most probable that Histogram Segmentation is being used here.
The histogram distribution of the values of all the pixels in the image is calculated. Then, the troughs in the graph are discovered (where fewer pixels of that colour exist), and this point is deemed to be the threshold. Any pixels with values over the threshold are grouped with the brighter pixels. Those pixels with values below the threshold are grouped with the darker pixels. A diagram of such a graph is in appendix 1.
For specific areas of the anatomy, a program may sometimes be able to label easily distinguished areas (generally by the pixel values and general shape). Other areas may be more ambiguous, with difficulty distinguishing the different pixel values. Human help can be enlisted here, with a technician picking out a group of pixels that clearly belong to a certain tissue. The software can then examine the other pixels in the image and assign them to the group that their value most closely matches.
In one of the systems I looked at (M.I.T. / G.E.M.S. image-guided surgery using MRI), the 'slices' from the imager were combined to create a 3-D model. This technique is not limited to image-guided systems, either; often it can help anatomical study to have 3-D computer models to examine. The pixels of the 'slices' are given Z-coordinates (based on the position of the 'slice' in the scan), and become full-dimensional voxels (volume elements).
This three dimensional voxel-based object may be rotated to
any desired angle using software, or the voxels may be
connected using flat faces or gouraud shading to render the
object at any desired quality. In the image-guided surgery
software, the scanned body part (head) is rotated to match
the patient's real head as seen from the surgeon's point of
view.
Of the three main internal medical imaging techniques discussed, there are definite differences between the effectiveness, cost and safety of the systems.
The x-ray picture has served the medical profession for more than a hundred years, but it has two major disadvantages: Firstly, the aspect of safety. It was not generally accepted that exposure to x-rays could be harmful until the early pioneers started to suffer the marks and scars of radiation, and die. Although proper precautions are taken nowadays by x-ray operators, and the dosages delivered are much smaller, it is still not very desirable to irradiate patients, and as a consequence, the operator is restricted in the number of plates they may expose. Secondly, x-rays are less effective than both CT scanners and MRI, since they only depict a two dimensional view of the patient from a certain angle.
The Computed Tomography scan is an improvement over the x-ray picture mainly because, like MRI, a series of 'slices' may be computed showing cross-sections of the patient. CT scanners have the advantage of being based on simpler technology than MRI systems (cheaper), but they still suffer from the problem of exposing the patient to doses of ionising radiation.
Magnetic Resonance Imaging systems are better at demonstrating anatomy than CT scanners, and also more sensitive to diseased tissue. Using MRI angiography allows pictures of flowing blood without the necessity of marker agents necessary to make veins show up on x-ray/CT scans. The patient is not exposed to any radiation during the scan. My personal opinion is that MRI systems are the best of the three techniques, however they are also very expensive through being technologically advanced.
Any CT scanner or magnetic resonance imaging system used to require the patient to lie full length inside the machine, whilst the scanner generated the images. This sometimes causes problems with people who are claustrophobic or very fat; either they will not or they cannot lie in the aperture. To some extent, this is now being remedied by the introduction of open-magnet MRI machines.
Computer generated imagery (whether two dimensional or three dimensional, real-time or preliminary) has proved itself invaluable in the field of modern medicine. The main advantage is that invasive surgery is unnecessary to find out the position, shape and healthiness of internal structures. A diagnosis can be made on the strength of the digital images, which are likely to give a clearer indication of details than surgery would in any case. The benefits of invasive surgery being avoided are obvious; surgery is always a risk to the patient's health, and not operating increases their chances of recovery. If surgery does prove necessary, then computer images/models will prove invaluable to the surgeon, allowing him to plan the operation in detail beforehand, and making the surgery quicker and more effective.
Finally, there are the general benefits of any kind of
digital imagery, in that the data is easily stored,
compressed, or transmitted to a colleague maybe hundreds of
miles away.
In terms of scope for future improvements in the
computer-assisted analysis of medical imaging, obviously a
computer program that was able to actually diagnose medical
problems from images would be a tremendous asset to the
medical profession, but realistically this kind of decision
is likely to continue to be made by doctors for the
foreseeable future.
In the not-so distant future is the impending design of
hardware and software capable of extending the image-guided
surgery techniques recently developed, to allow real-time
rebuilding of the three dimensional model which assists the
neurosurgeon during surgery.
The scope of the possible applications for image-guided
surgery in the future is enormous; in radiotherapy, for
instance, the radiotherapist can ensure that the beams of
radiation converge accurately at the desired anatomical site,
with minimal damage to surrounding tissue. The size and
position of tumors may also be easily reviewed before and
after radiation or drug treatment. Investigation has already
commenced into whether three dimensional representations
built from MRI scans might be more effective at early
detection of breast cancer than mammograms.
There are still considerable challenges for software authors
in the field of real-time updated image-guided surgery.
Considerable problems still exist with creating
representations of moving organs; models can quickly become
inaccurate when the patient breathes. Algorithms are
currently under development to try to predict tissue
deformations in such cases.
http://www.imaginis.net/ct-scan/
http://www.cis.rit.edu/htbooks/mri/inside.htm
http://www.eos.ncsu.edu/bae/research/blanchard/www/465/textbook/imaging/projects/MRI/
| Title | Author/s | ISBN/Periodical | Medium |
| Machine Perception | Nevati | 0135419042 | Book |
| Image Guided Surgery | Eric,Grimson,Kikinis,Jolesz,Black | Scientific American | Article |
| Computer Vision | Boyle,Thomas | 0632015772 | Book |

Histogram Segmentation
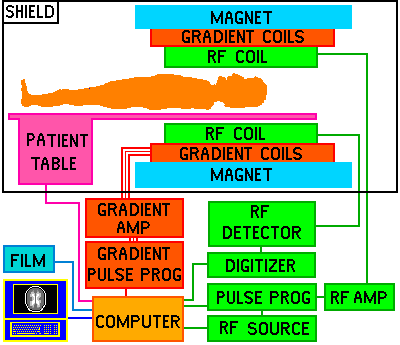
Block diagram of MRI scanner